|
A teacher may use a metal/nonmetal scheme for an introductory lesson.With the flexibility of digital periodic tables, it is easy to switch between color-coding systems depending on what property needs emphasis. Colors can demonstrate this gradient clearly. Atomic RadiusĪtomic radius tends to decrease moving left to right across a period. Groups or families with similar properties are easier to locate when coded by color (such as the noble gases or halogens). Electron ShellsĮlements with equal numbers of electron shells can be colored alike to illustrate shell filling patterns. ElectronegativityĪ color gradient from least electronegative (like francium) to most electronegative (like fluorine) visualizes this important periodic trend. Colors can group elements with equal valence electron counts. The number of valence electrons is an important factor in chemical bonding. Color-coding them makes those domains clear at a glance. Metals, nonmetals, and metalloids occupy very different regions of the table. Some of the key elemental properties and periodic trends that different color schemes emphasize include: PhasesĬolor-coding elements by their state at standard temperature and pressure (usually solids, liquids, and gases) shows the distribution of phases and highlights anomalies like mercury as a liquid metal. Overall, the color visualization transforms the periodic table from a basic chart to an informative, useful tool for analysis and understanding. Engages students and makes the table less abstract and more approachable.Demonstrates the basic organization principles of the table.Distinguishes groups and periods at a glance.Highlights trends like electronegativity, electron shells, and reactivity.Allows quick differentiation of metals, nonmetals, and semimetals.Improves memorization and recall of elemental properties and categories.Makes patterns and relationships between elements more visible.Electron Configuration: Colors group elements by common electron orbital patterns.Ĭolor-coding the periodic table offers many benefits for students, teachers, and researchers alike:.Electronegativity: A gradient scale visualizes increasing electronegativity values.Phases: Colors indicate phase at room temperature – solids, liquids, and gases.Blocks: The s-, p-, d- and f-blocks can be colored distinctly to visualize patterns.Periods: Elements in the same period or row can be colored identically to show trends.Groups/Families: Elements in the same group or family are colored identically, like halogens in light green.Metals, Nonmetals, and Metalloids: Metals are often colored orange/yellow, nonmetals green, and metalloids a combination.Here are some of the most common ways color is used to differentiate elements in modern periodic tables: While there is no universally accepted standard, certain color schemes have become more widely used, like highlighting metals in yellow/orange, nonmetals in green, noble gases in purple, alkali metals in pink, alkaline earth metals in blue, and halogens in light green. Today, many varieties of color-coded periodic tables exist. Subsequent chemists introduced additional color schemes to illustrate properties like electronegativity, electron configuration, and more. In the 1950s, American chemist Edgar Fahs Smith created one of the earliest color-coded periodic tables, using colors to categorize metals, nonmetals, and inert gases. The idea emerged in the 20th century as chemists looked for ways to better visualize the relationships between elements. History of Color Coding the Periodic TableĪlthough Dmitri Mendeleev is credited with creating the first periodic table in 1869, it did not originally contain any color coding. 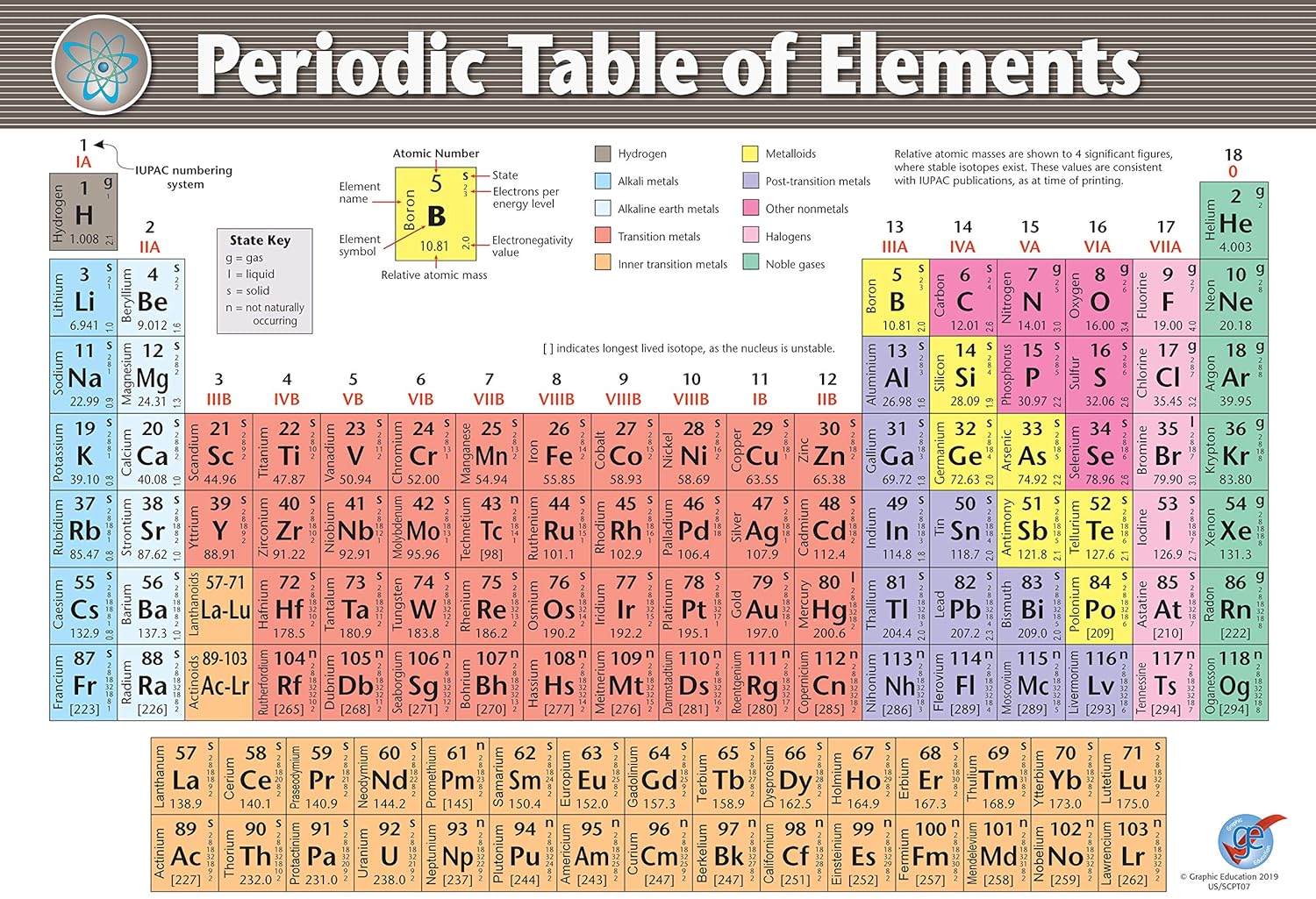
While the standard black-and-white version is familiar to most, color-coded periodic tables have become increasingly popular for visualizing different properties and trends of the elements. The periodic table is one of the most recognizable scientific tools, displaying all the known chemical elements organized by their atomic number.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
